What modern neuroscience tells us about posture and gait
A scientific review by Takusaki et al. 2024 in Ageing and Neurodegenerative Diseases, shows that human posture and gait are not controlled by a single structure or system but emerge from a distributed neural network spanning the cerebral cortex, basal ganglia, cerebellum, brainstem, and spinal cord. While muscles and joints provide the mechanical means for movement, the organization, timing, and stability of walking are fundamentally neural achievements shaped by evolution, development, and aging.
At the core of locomotion lies a conserved vertebrate locomotor system. Across species—from fish to humans—locomotion is generated by spinal central pattern generators (CPGs) that produce rhythmic limb movements. These spinal networks are activated and modulated by brainstem locomotor regions, which send descending signals through reticulospinal pathways to regulate rhythm, coordination, and postural muscle tone. This “automatic” system allows walking to occur with little conscious awareness under stable conditions.
Human upright walking, however, places far greater demands on this core system. Bipedal posture requires continuous regulation of balance, muscle tone, and alignment against gravity. To meet these demands, humans rely on additional supraspinal control, particularly from cortical, basal ganglia, and cerebellar systems. These higher centers do not replace the brainstem–spinal machinery; instead, they shape and adapt it to context, intention, and environmental complexity.
A key finding across posture and gait research is the importance of anticipatory postural adjustments (APAs). Before a step, turn, reach, or change in direction, the nervous system generates feedforward postural commands that prepare the body for the upcoming movement. APAs depend on intact communication between the cerebral cortex, brainstem, and spinal cord. When APAs are delayed, reduced, or poorly scaled, gait becomes unstable even if muscle strength is preserved.
Sensory integration plays a central role in this system. Visual, vestibular, proprioceptive, and tactile inputs continuously update posture and gait control. Vestibular signals, in particular, are critical for maintaining upright orientation relative to gravity. These sensory signals are processed in parietal and temporal cortical regions to generate a coherent perception of body position in space, which is then relayed to frontal motor regions for action planning.
The frontal cortex, especially prefrontal and premotor areas, contributes to posture and gait by enabling goal-directed behavior, attention, task switching, and multitasking. Walking in the real world rarely occurs in isolation; it often involves navigating obstacles, changing direction, or performing cognitive tasks simultaneously. Research consistently shows that as cognitive load increases, gait performance deteriorates—particularly in older adults and individuals with neurological disease.
The basal ganglia are essential for selecting, initiating, and automating gait patterns. Dopaminergic signaling allows movements to become efficient and habitual. When basal ganglia output is disrupted, as in Parkinson’s disease, gait loses automaticity, leading to slow initiation, freezing, and difficulty switching between movement patterns. Importantly, basal ganglia output also influences brainstem locomotor regions directly, linking motivation and movement execution.
The cerebellum contributes through error detection, timing, and adaptation. By comparing intended movement with sensory feedback, the cerebellum updates internal models that refine posture and gait. This allows humans to adjust walking patterns to uneven terrain, narrow spaces, or novel environments. Cerebellar dysfunction leads to gait variability, poor coordination, and reduced adaptability, even when strength is intact.
Aging introduces structural and neurochemical changes across this network. Brain volume reduction—especially in frontal and temporal regions—declines in white matter integrity, and degeneration of broad-projecting neurotransmitter systems (dopamine, acetylcholine, noradrenaline, serotonin) all reduce the efficiency of posture–gait control. As a result, gait becomes slower, more variable, and increasingly dependent on conscious attention. Dual-task walking and obstacle negotiation become disproportionately challenging.
Neurodegenerative diseases amplify these age-related effects in distinct ways. Alzheimer’s disease affects executive and attentional systems, increasing gait disruption under cognitive load. Parkinson’s disease primarily disrupts basal ganglia–brainstem interactions, impairing initiation and automaticity. Progressive supranuclear palsy damages brainstem and cortical connections critical for balance and gaze control, leading to early falls. Across these conditions, posture and gait disturbances reflect network-level failure, not isolated muscle or joint pathology.
Collectively, research supports a unified conclusion: human posture and gait are emergent properties of a multi-level neural system. Stable upright walking depends on the integration of automatic spinal–brainstem mechanisms with higher cortical planning, sensory prediction, emotional regulation, and adaptive learning. Disruption at any level—sensory, cognitive, emotional, or motor—can manifest as postural instability or gait dysfunction.
For therapists, this body of research reinforces the importance of viewing posture and gait through a neuro-integrative lens. Observable changes in posture or walking pattern often reflect altered neural coordination rather than purely local musculoskeletal deficits. Effective rehabilitation therefore benefits from combining physical conditioning with context-rich, task-specific, and cognitively informed movement training that targets the nervous system as much as the tissues it controls.
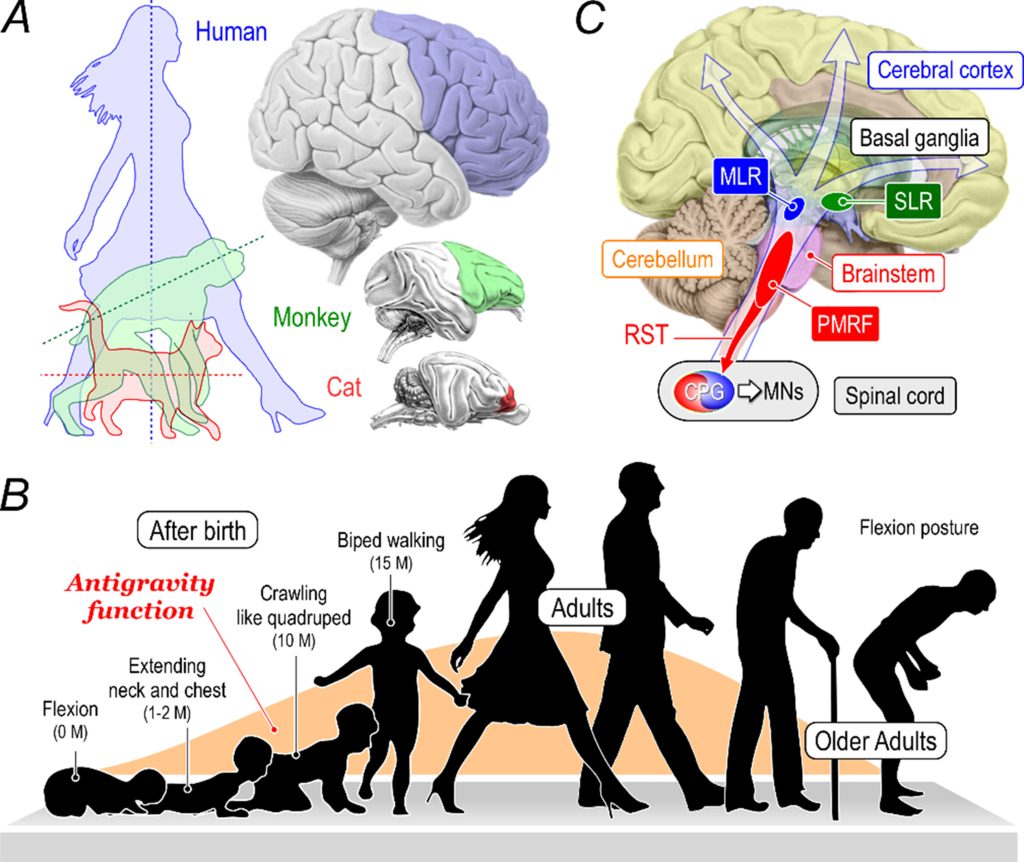
Takusaki et al. 2024: Evolution and development of humans.
(A) Evolutional changes in posture and brain in mammals. Through vertebrate evolution, the body axis of animals such as cats (red), monkeys (green), and humans (blue) changed from horizontal to vertical, and the frontal lobe area in each animal’s cerebral cortex expanded considerably;
(B) Changes in posture and gait with postnatal development and aging of humans. The antigravity function, indicated by the orange color, is facilitated by postnatal development, but declines due to aging. While humans exhibit a flexion posture immediately after birth, their antigravity ability develops over time. Specifically, at 1-2 months, the baby extends its neck and chest, and at around ten months, it begins to crawl. Then, at approximately 15 months, the baby starts to walk upright on two legs. Human antigravity capacity reaches its peak during adulthood. However, antigravity capacity declines with age, manifesting as a decreased walking speed and a flexed posture in old adults;
(C) An open arrow indicates that postnatal brain maturation progresses from the lower to the upper brain areas. Specifically, spinal reflex neural circuits, including the CPGs, can be functional at birth. Then, the neural networks between the brainstem and spinal cord, including the PMRF and the RST, are maturated. The upper brainstem includes neural structures involved in innate motor functions such as swallowing, vocalization, and excretion, in addition to those contributing to the automatic posture-gait control, including the MLR and SLR. Maturation further progresses to involve the subcortical structures (the basal ganglia, limbic system, and thalamus), cerebellum, and cerebral cortical structures, operating higher brain functions.
CPGs: Central pattern generators; PMRF: pontomedullary reticular formation; RST: reticulospinal tract; MLR: midbrain (mesencephalic) locomotor region; SLR: subthalamic (diencephalic) locomotor region; MNs: motoneurons.
Takakusaki, K., Takahashi, M., Kaminishi, K., Fukuyama, S., Noguchi, T., Chiba, R., & Ota, J. (2024). Neural mechanisms underlying upright bipedal gait: role of cortico-brainstem-spinal pathways involved in posture-gait control. Ageing and Neurodegenerative Diseases, 4(3), N-A.


